- Blog
- Web Data Extractor Pro 2.3 Crack
- Free Download Mp3 Song Papi Chulo Remix
- Captive Market Game
- Garbh Sanskar Book In Hindi Pdf
- Romancing Saga Minstrel Song Rom
- Download Mp3 Lagu Arab Umi Kulsum
- Uplay_r1_loader.dll Assassins Creed Black Flag
- Bryan Magee The Great Philosophers Pdf To Word
- Download Film Captain America The Winter Soldier Sub Indo Bluray
- Toad For Oracle License Key And Site Message
- Play Aqua Energizer Hacked
- Beamng.Drive 0.4.0.6
- Swords And Sandals 3 Full Version Download Crack Fifa
- Samsung I9100 Efs Repair File
- Adobe Indesign Cs6 Full En Mega
- Photomatix Pro Mac Torrent Pirate
- Optical Flares Keygen Download Sony
- Social Club V1.1.0.6 Setup.exe
- Aplikasi Visual Foxpro Gratis
- E Aadhaar Card By Enrollment Number
- Kakasoft Folder Protector Pro V6.30
- Haldor Topsoe Hydrocracking
- Ibps Study Material Pdf For Ibps Clerk Po Exam 2013
- Download Fated To Love You Sub Indonesia
- Nc Code Simulator
- Internet Tv Packages
- Samsung Pvr Drm Decrypter
- Amtlib.dll Adobe Premiere Cc 2015 Copy File
- Sheetcam Tng License
- Monsanto Tensometer Manual
- Crestron Simpl Software Download
- Barber Chair Repair Manual
- Red Love Is All Around Rar Download
- Aromaticity In Benzenoid And Non Benzenoid Compounds Pdf
- Download Film Love Of Siam Sub Indo
- Torrent Janome Digitizer Pro
- Armata Mortii Joc Romanesc Torent
A PDF file should load here. If you do not see its contents the file may be temporarily unavailable at the journal website or you do not have a PDF plug-in installed and enabled in your browser.
Alternatively, you can download the file locally and open with any standalone PDF reader:
Aromaticity of benzenoid hydrocarbons with inserted –B=B– and –BH–BH– groups: a comparison
Aromaticity of benzenoid hydrocarbons with inserted -B=B- and -BH-BH- groups: a comparisonKrzysztof K. Zborowski 0 1 2Ibon Alkorta 0 1 2Jose Elguero 0 1 20 Instituto de Qu ́ımica Me ́dica (C.S.I.C.) , Juan de la Cierva 3, 28006 Madrid , Spain1 Faculty of Chemistry, Jagiellonian University , 3 Ingardena Street, 30-060 Krako ́w , Poland2 & Krzysztof K. ZborowskiStructures of selected polycyclic conjugated hydrocarbons with -B=B- and -BH-BH- moieties inserted in different places were calculated at the B3LYP/6311??G** level and their aromatic properties evaluated. HOMA, NICS(0), NICS(1)zz, K and PDI indices were used for studying their aromatic properties. Both optimized planar (as in parent hydrocarbons) and non-planar structures were taken into account. It is shown that insertion of both types of boron groups disturbs and decreases the aromaticity of the corresponding hydrocarbons. The decreasing effect of the -BH-BH- group is much stronger. What is quite intriguing is that it appears that non-planar structures of the studied compounds have a little higher aromaticity than the strictly planar ones. Mutual correlations between results obtained by different aromaticity indices are calculated and thoroughly discussed.Aromaticity; Substituted benzenoid hydrocarbons; Boron compounds; HOMA; NICSIntroductionReplacement of one or more carbon atom(s) in benzene andin polycyclic aromatic hydrocarbons (PAH) byheteroatoms other than the typical ‘‘heterocyclic’’ elements likenitrogen, oxygen, phosphorus and sulfur has been recentlya topic of several studies [1–6]. Such structuralmodifications are important because they can create compoundswith unusual properties. The preparation of the (poly)cyclichydrocarbons doped by boron(s) discussed in the presentwork would be difficult. However, some publicationsreport the syntheses of other types of hydrocarbons withinserted BB bonds [7–12]. Very important for furtherdevelopment of the chemistry of substituted boron(s)hydrocarbons is the understanding of the effects of thereplacement of C by B- [13, 14].Among others, introduction of boron into a carbon placein the hydrocarbon skeleton provides the possibility ofobtaining new materials with potentially useful properties.Boron is one of the key elements [15]; it forms enormousnumber of compounds, and the riches of its chemistry canbe compared only with chemistries of such basic elementslike carbon, silicon, nitrogen or oxygen. The main groupsof boron compounds are borates [16] and borosilicates[17]. Many compounds built from boron and hydrogenatoms (boranes), and from boron, hydrogen and carbonatoms (carboranes) are also well known [18].As indicated, even in the commonly used name of‘‘aromatic hydrocarbons,’’ the aromaticity is one of themost significant properties defining these compounds. Thearomaticity defines their structure as well as theirreactivity. Thus, increasing or decreasing the aromatic propertiesin substituted hydrocarbons is very important for theirproperties and potential applications. Therefore, theinfluence of heteroatoms on the aromaticity of conjugatedhydrocarbons is a subject worth studying.In this work, we examined the aromatic properties ofseveral aromatic hydrocarbons, such as benzene,naphthalene, anthracene, phenanthrene, pyrene and coronene,substituted by –B=B– and –BH–BH– moieties in variouspositions. Recently we have published two papers in whicharomaticity of some conjugated hydrocarbons doped by asingle boron atom (–B=) [19] and by two boron atoms–B=B– [20] was briefly studied. Those publicationsresulted in arising the question of what will happen concerningsubstitution by the –BH–BH– group [21], something thatcannot be predicted. Thus, we decided to study in moredetail the aromaticity of hydrocarbons with structuresdisturbed by replacing a CC bond by –B=B– or –BH–BH–groups.The question of the aromatic properties of compoundscontaining boron is not new. Borazine, B3N3H6, the mostfamous member of the azaborines group, is even named‘‘inorganic benzene’’ [22]. Aromaticity of other boroncompounds was also reported several times [23–28]. Thus,we think that investigation of the aromatic properties ofboron-substituted hydrocarbons and the evaluation of theusefulness of different aromaticity indices for thedescription of such compounds’ aromaticity was a reallyinteresting topic.Computational detailsManifestations of strong cyclic electronic delocalization,commonly known under the little confusing name of‘‘aromaticity,’’ are an intriguing but still not fullycharacterized phenomenon. Its definition is still under vividdisputation [29], and as a result of this ambiguously definition,there is no single method that is generally accepted foraromaticity strength evaluation. So-called aromaticityindices are a set of very different methods based on threemain aromaticity criteria: energetic, geometric andmagnetic [30], as well as electronic indices that derive aromaticproperties directly from molecular wave functions analysis[31]. It is recommended to use more than one aromaticityindex of aromaticity in order to consider all aspects ofaromaticity [32, 33].That is why, in order to obtain a wide range ofinformation about aromatic properties of the structures studiedin this paper, we will use different sets of aromaticityindices, namely: harmonic oscillator model of aromaticity(HOMA) [34], nucleus-independent chemical shift (NICS)[35, 36], para delocalization index (PDI) [37] and magneticsusceptibility (K) [38]. Parameters of the HOMA model(optimum aromatic bond length Ropt and normalizationconstant a) for the CC (Ropt = 1.388 A˚ , a = 257.70), BC(Ropt = 1.4378 A˚ , a = 118.009) and BB (Ropt = 1.5665A˚ , a = 244.147) bonds were taken from references [19, 20,39]. Values of the delocalization indices [40] betweenatoms in para position, necessary for PDI calculations,were obtained with the AIMAll package [41]. Indices usedin this work can be divided into two groups. HOMA, NICSand PDI indices were used to determine aromaticproperties of all single rings of studied structures. On theother hand, HOMA (again) and K provided the totalaromaticity of the compounds.The B3LYP [42] /6-311??G(d,p) [43] level ofcomputation was used in this work. This level of theory hadbeen used in our previous papers for calculations of thestructures of boron-substituted hydrocarbons [19, 20].Thus, such a basis set enabled the direct comparisonbetween the results obtained in this work and thosepublished previously. Strictly planar (like for the parenthydrocarbons) and relaxed unplanar structures wereexamined. All structural optimizations were executed usingthe Gaussian’09 Revision A.02 package [44].Results and discussionAll compounds studied in this work obtained bysubstituting the CC bonds in cyclic conjugated hydrocarbons by–B=B– or –BH–BH– groups are presented in Fig. 1,compounds 1 to 12; series a corresponds to –B=B–derivatives and series b to –BH–BH– ones. Ring labeling isalso provided.Geometry optimization procedure was performed for allcompounds. In general, insertion of boron fragments intothe flat hydrocarbon’s structures results in destroying theplanarity of the ring. Thus, the boron derivatives of thestudied hydrocarbons are not planar. The CBBC dihedralangle in boron compounds is about 40 for a series and 30for the b series. Due to the non-planar structures of thecompounds, we decided to determine the aromaticities ofboron derivatives with a plane of symmetry (like in theparent hydrocarbons) and without it (like in relaxedstructures of boron derivatives). There are two exceptionsfrom this typical behavior for boron-substitutedhydrocarbons, in both cases for –BH–BH– substituted derivatives.The structure of compound 10b is flat; no loss of planarityis observed. On the other hand, in compound 5b, theboroncontaining ring transforms, upon optimization, into apyramidal structure with a five-membered ring (fourcarbons and a BH group) in the base of the pyramid and asecond BH group at the top. Due to its unusual structure,this ‘‘ring’’ has been excluded from further analyses. For allother than 5b and 10b compounds, the planar structures aretransition states between two equivalent non-planarminima. The energy barriers between planar and non-planarstructures are different, from about 0.1 kJ mol-1 (forcompounds 2a, 9a and 12b) up to about 43 kJ mol-1 forcompound 5a.Aromaticity data for the whole structures ofboronsubstituted hydrocarbons will be presented first. Theanalysis is based on the values of the HOMA index for allbonds (HOMAtotal) as well as on values of magneticFig. 1 Molecular structures andindividual ring labeling of thestudied hydrocarbons withinserted –B=B– groups, a series,and inserted –BH–BH– groups,b series. Hydrogen atomsconnected to the carbon atomsare omitted for claritysusceptibility (K). Data obtained by these two methods arecollected in Table 1 and presented in Fig. 2.Variations of the HOMAtotal index for hydrocarbons ofthe a series are surprisingly small. Most HOMAtotal valuesfor this group of compounds are between 0.67 (compound9a) and 0.53 (compound 5a). Thus, all these compoundscan be classified as moderate aromatic. Only compound 1a(the benzene derivative) has higher HOMAtotal value (0.79)and from this point of view can be described as almosthighly aromatic. Also the aromaticity of compound 3a(HOMA = 0.68) is quite high. On the other side,HOMAtotal values for the compounds of the b series spanover a much larger region. They are antiaromatic(compounds 1b and 3b) or non-aromatic (2b, 7b, 8b, 9b, 11b);small aromatic properties are suggested by the HOMAtotalvalues for compounds 4b, 5b, 6b, 10b and especially 12b.The last compound can be considered as moderatearomatic. However, its moderate aromaticity is due to the factthat compound 12b, a derivative of coronene, is muchlarger than the other studied systems. Thus, boronsubstitution strongly disturbs electronic structure probably onlyin a part of its carbon skeleton. The biggest difference inaromatic properties is observed between benzenederivatives of both series. One (benzene with a –B=B– group, 1a)is aromatic, while strong antiaromaticity is observed for its–BH–BH– counterpart, 1b. Benzene is the smallerhydrocarbon considered in this work, so it is reasonable that inthis case, introduction of the boron atoms can changecompletely its electronic structure. Differences betweenHOMAtotal values for planar and non-planar structures ofa series are always small or very small. Bigger changesbetween planar and non-planar systems are observed forthe b series (up to -0.83). What is intriguing is thatHOMAtotal values are usually a bit higher for relaxednonplanar structures. This behavior is in opposite to the purehydrocarbons where destroying the planar structure resultsin decreasing aromatic properties [45].In general, calculated K values follow HOMAtotal data,i.e., compounds of the a series are more aromatic than theirb series counterparts. However, differences betweenmagnetic susceptibilities of the same compound in its planarand non-planar structures are much greater than for theHOMAtotal results. In addition, magnetic susceptibilitycalculations afforded sometimes quite unexpected values.Such an unexpected case is compound 2, wheresignificantly more negative values are predicted for thehydrocarbon with the –BH–BH– insertion, 2b. Anothersomewhat strange case is that of compound 4. For thiscompound, a very huge, difficult to explain, change inmagnetic properties occurred during transition from the flatto the relaxed structure without symmetry plane. Such ahuge change is not observed for any other structurereported in this work. The origin of such unexpectedartifact in the magnetic susceptibility data is worth of a futuremore detailed study.Low correlations are observed between HOMAtotal andK data presented in Table 1. For both series, correlationcoefficients between HOMAtotal and K are almost zero.Such a result can support the thesis about themultidimensional character of aromaticity, where structural andmagnetic indices correspond to different manifestations ofthis property [32]. What is even more intriguing is thatthere is no correlation between magnetic susceptibilities(K) determined for the –B=B– substituted compounds andtheir –BH–BH– analogs (correlation coefficients, c.c.,equal to -0.07 for planar and -0.11 for non-planarstructures). At the same time, there is some correlationbetween HOMAtotal data for two groups of boron-dopedhydrocarbons, correlation coefficients being 0.46 and 0.66for planar and non-planar structures, respectively.Now the aromatic properties of individual rings in thestructures of boron-substituted hydrocarbons will bediscussed. The data are collected in Table 2 (for compoundscontaining –B=B– group, 1) and in Table 3 (compoundswith the –BH–BH– group, 2). Figure 3 reports thearomaticity data for all rings with boron atoms.It should be mentioned at this moment that there aresome problems with using some standard aromaticityindices for the compounds containing boron atoms. First ofall, NICS index data look sometimes unreliable for theserings. This can be the result of the fact that electrons closeto B atoms are freer to move than those of C atoms andcreate ring currents that produce these high NICS(0)values. The fact that electrons close to B atoms are morediffuse can influence also the PDI data, due to the highervalues of para delocalization indices obtained in theserings. Thus, PDIs for rings containing B atoms are likely tobe somewhat overestimated.HOMA values for the individual rings with only carbonatoms are usually quite ‘‘stable’’ during ‘‘transition’’ fromplanar to non-planar structures. Some changes appear forthe rings that have a boundary with the ring containingboron atoms. For rings with boron atoms, transforming themolecular structure from planar to non-planar resultssometimes in a substantial change. It can be noticed that innon-planar structures, some rings containing boron atoms,compounds 3a and 6a as well as 1b, 2b and 3b, switch theirantiaromatic properties into non-aromatic ones. Almost allrings increase their HOMA values in non-planar structures,and this is the rule for all the rings with boron atoms. Theexception from this rule occurs only for benzene derivative1a. There are no dramatic changes after the plane of thesymmetry disappears in the case of the PDI data. HigherPDI values are observed for the non-planar structures foralmost all rings with only carbon atoms and for all ringscontaining boron (even for 1a). A different behavior isobserved for the NICS(0) and NICS(1)zz models ofaromaticity evaluation. In these cases, it is difficult to predictwhat is more probable is decreasing or increasing inaromatic properties in non-planar structures in comparisonwith the planar ones. In addition, sometimes very hugevalues, both very positive and very negative, are predictedby the NICS calculations.In general, more significant perturbations of aromaticproperties are observed in the rings with boron atoms than inthose consisting of only carbon atoms. Depending on theposition of the carbocyclic ring in the structures of thecompounds, either increasing or decreasing in the aromaticproperties can be observed. In general, aromatic properties ofpure carbon rings that border with carbon–boron ones aremore affected (decreased) than those located far from borongroups. It is difficult to decide about the way of aromaticproperties affecting of pure carbon rings directly connectedwith carbon–boron rings. There is a very variablerelationship between aromaticity and the structure of the studiedhydrocarbons. For some structures, aromaticity of such ringsis higher in hydrocarbons with the –B=B– group (a series),and for others, the opposite effect is observed.We will examine now the rings in which boron atomsare present. Insertion of the –B=B– group into thePDIhydrocarbons structures results in carbon–boron rings withdifferent aromatic properties. Most of them are moderatearomatic. However, in some rings, non-aromatic orantiaromatic properties are calculated. As it was mentionedabove, aromatic properties of the boron-containing ringsincrease in their non-planar structures. On the contrary,insertion of the –BH–BH– group, b series, leads to obtaincarbon–boron rings with clearly antiaromatic properties(negative HOMA, positive NICS(0) and NICS(1)zz as wellas low PDI values); see Table 3.As it was mentioned earlier, not always different indicesof aromaticity describe aromatic properties of chemicalspecies in the same way. Thus, it is interesting to checkwhether their results correlate or not in such a difficult caseas the aromaticity of rings with boron atoms in structures ofPAHs.An attempt to find mutual correlations between differentindices describing the same set of compounds (substitutedhydrocarbons assumed as planar structures, substitutedhydrocarbons in their relaxed non-planar structures or boththese groups together) provides following results. Veryhigh correlation (c.c. 0.99) is observed between NICS(0)and NICS(1)zz data in planar structures (NICS(1)zz datawere calculated only in this case). Significant correlation isnoticed also between HOMA and PDI for b series and allstudied substituted hydrocarbons in planar structures(correlation coefficients higher than 0.80). Unfortunately thiscorrelation does not exist for planar –BH–BH– species.Weak correlations (c.c. from the range 0.4–0.6) areobtained for some groups of compounds between HOMA–PDI and NICS–PDI results. Only small correlations areobserved between HOMA and both NICS indices.We have also checked how the results provided by thesame indices for carbon–boron rings in both groups ofsubstituted hydrocarbons correlate. The results are ratherpoor, i.e., in most cases, very low correlations are found.Contrary to this general trend, strong correlation (c.c. 0.97)is observed between HOMA data for rings with –B=B– and–BH–BH– groups in non-planar structures. This result isinteresting because in a recent paper [46], it was shown thatin polycyclic conjugated hydrocarbons, in which twocarbon atoms are replaced by various pairs of heteroatoms,peripheral lengths of the heteroatomic bonds are strictlycorrelated with the corresponding CC bond lengths in thenative hydrocarbons. However, in the mentioned paper,only planar structures were considered. HOMA index, asother structural indices of aromaticity, is based on bondlengths. Thus, it was interesting to check whether there is acorrelation between the BB bond lengths in both series ofcompounds. The result is curious. There is a strongcorrelation (c.c. -0.99) between BB bond lengths in planarstructures, while there is lack of correlation (c.c. -0.03) innon-planar ones. Thus, there is the strong correlationbetween HOMA data and no correlation between BB bondlengths for non-planar carbon–boron rings, and theopposite situation occurs for these rings in planar structures.ConclusionsStructures of several PAHs with inserted –B=B– or –BH–BH– groups, series a and b, have been investigated. Wehave clearly shown that insertion of both types of borongroups disturbs and decreases aromaticity of the precursorhydrocarbons. However, the decreasing influence of the –BH–BH– group is much stronger and leads in all cases tocarbon–boron rings with antiaromatic properties. If onechanges C–H by B in a benzenoid structure, the number ofvalence electrons available for r and p bonding does notchange and as a result, the change of –CH=CH– by B=B inbenzene leads to a 6-p-electron system which in principleshould be aromatic according to Hu¨ckel’s rule. On theother hand, if one changes C–H by B–H, then one electronless is present for r and p bonding, and since r is preferredover p bonding, then the change of –CH=CH– by –BH–BH– reduces the number of available p-electrons in asubstituted benzene ring to 4 and this should lead toantiaromatic species. Thus, it is reasonable that aromaticityshould disappear in –BH–BH– derivatives.Insertion of the –B=B– moiety gives rings with differentaromatic properties. Antiaromatic, non-aromatic, slightlyaromatic rings and even one with quite strong aromaticitywere observed. What is quite unexpected is that non-planarstructures have a bit higher aromaticity than strictly planarones. This is observed despite the fact that parenthydrocarbons, as well as some boron clusters in whicharomaticity was postulated, are planar. It is possible, even if itsounds quite strange, that in hydrocarbons substituted byboron groups, the aromaticity is a driving force for therings to lose their planarity.Acknowledgments We thank the Ministerio de Ciencia eInnovacio´n (Project No. CTQ2012-35513-C02-02) and the ComunidadAuto´noma de Madrid (Project MADRISOLAR2, ref.S2009/PPQ1533) for their continuing support. Thanks are also given to ‘‘ICM’’Warsaw (Project Number G17-8) and to the CTI (CSIC) computercenters for an allocation of computer time.Open Access This article is distributed under the terms of theCreative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use,distribution, and reproduction in any medium, provided you giveappropriate credit to the original author(s) and the source, provide alink to the Creative Commons license, and indicate if changes weremade.1. Omelchenko IV , Shishkin OV , Gorb L , Leszczynski J , Fias S , Bultinck P ( 2011 ) Chem Phys Phys Chem 13 : 20536 - 205482. Dou C , Saito S , Matsuo K , Hisaki I , Yamaguchi S ( 2012 ) Angew Chem Int Ed 51 : 12206 - 122103. Roszak R , Roszak S ( 2015 ) J Mol Model 21 : 284. Sa ´nchez-Sanz G ( 2015 ) Tetrahedron 71 : 826 - 8395. Karabiyik H , Sevinc¸ek R , Karabiyik H ( 2015 ) J Phys Org Chem 28 : 490 - 4966. Tejchman W , Proniewicz LM , Zborowski KK ( 2015 ) J Phys Org Chem 28 : 536 - 5417. Moezzi A , Bartlett RA , Power PP ( 1992 ) Angew Chem Int Ed 31 : 1082 - 10838. Moezzi A , Olmstead MM , Power PP ( 1992 ) J Am Chem Soc 114 : 2717 - 27199. Grigsby WJ , Power PP ( 1996 ) Chem Commun 1996 : 2235 - 223610. Grigsby WJ , Power PP ( 1997 ) Chem Eur J 3 : 368 - 37511. No¨th H , Knizek J , Ponikwar W ( 1999 ) Eur J Inorg Chem 1999 : 1931 - 193712. Bissinger P , Braunschweig H , Damme A , Kupfer T , Vargas A ( 2012 ) Angew Chem Int Ed 51 : 9931 - 993413. Poater J , Sola ` M, Vinas C , Teixidor F ( 2013 ) Chem Eur J 19 : 4169 - 417514. Poater J , Sola ` M, Vinas C , Teixidor F ( 2014 ) Angew Chem Int Ed 53 : 12191 - 1219515. Pauling L , Pauling P ( 1975 ) Chemistry . W. H. Freeman and Company, San Francisco16. Wiberg E , Holleman AF ( 2001 ) Inorganic chemistry . Elsevier, Amsterdam17. Vogel W ( 1994 ) Glass chemistry . Springer-Verlag GmbH, Berlin18. Cotton FA , Wilkinson G , Gaus PL ( 1987 ) Basic inorganic chemistry . Wiley, New York19. Zborowski KK , Alkorta I , Elguero J , Proniewicz LM ( 2012 ) Struct Chem 23 : 595 - 60020. Zborowski KK , Alkorta I , Elguero J , Proniewicz LM ( 2013 ) Struct Chem 24 : 543 - 54821. Ponikvar-Svet M , Zeiger DN , Keating LR , Liebman JF ( 2013 ) Struct Chem 24 : 2101 - 211422. Boese R , Maulitz AH , Stellberg P ( 1994 ) Chem Ber 127: 1887 - 188923. Aihara J , Kanno H , Ishida T ( 2005 ) J Am Chem Soc 127 : 13324 - 1333024. Yu HL , Sang RL , Wu YY ( 2009 ) J Phys Chem A 113: 3382 - 338625. Del Bene JE , Yanez M , Alkorta I , Elguero J ( 2009 ) J Chem Theory Comput 5 : 2239 - 224726. Kiran BK , Kumar GG , Nguyen MT , Kandalam AK , Jena P ( 2009 ) Inorg Chem 48 : 9965 - 996727. Ruman T , Jarmuła A , Rode W ( 2010 ) Bioorg Chem 38 : 242 - 24528. Qiang C , Hui B , Jin-Chang G , Chang-Qing M , Si-Dian L ( 2011 ) Phys Chem Chem Phys 13 : 20620 - 2062629. PvR Schleyer, Jiao H ( 1996 ) Pure Appl Chem 68 : 209 - 21830. Krygowski TM , Cyran´ski MK, Czarnocki Z , Ha¨felinger G , Katritzky AR ( 2000 ) Tetrahedron 56 : 1783 - 179631. Poater J , Dura´n M , Sola` M, Silvi B ( 2005 ) Chem Rev 105 : 3911 - 394732. Cyran´ski MK, Krygowski TM , Katritzky AR , Schleyer PvR (2002) J Org Chem 67 : 1333 - 133833. Feixas F , Matito E , Poater J , Sola` M (2007) J Phys Chem A 111: 4513 - 452134. Kruszewski J , Krygowski TM ( 1972 ) Tetrahedron Lett 13 : 3839 - 384235. PvR Schleyer, Marker C , Dransfeld A , Jiao HJ , Hommes NJRV ( 1996 ) J Am Chem Soc 118 : 6317 - 631836. Corminboeuf C , Heine T , Seifert G ( 2004 ) Schleyer PvR . Phys Chem Chem Phys 6 : 273 - 27637. Poater J , Fradera X , Duran M , Sola` M (2003) Chem Eur J 9: 400 - 40638. Dauben HJ Jr, Wilson JD , Laity JL ( 1968 ) J Am Chem Soc 90 : 811 - 81339. Krygowski TM ( 1993 ) J Chem Inf Comput Sci 33 : 70 - 7840. Bader RFW ( 1990 ) Atoms in molecules: a quantum theory . Oxford University Press, Oxford41. AIMAll, Version 11 . 10 .16, Keith TA ( 2011 ) TK Gristmill Software, Overland Park (aim .tkgristmill.com)42. Becke AD ( 1993 ) J Chem Phys 98 : 5648 - 565343. Krishnan R , Binkley JS , Seeger R , Pople JA ( 1980 ) J Chem Phys 72 : 650 - 65544. Gaussian 09, Revision A . 02 , Frisch MJ , Trucks GW , Schlegel HB , Scuseria GE , Robb MA , Cheeseman JR , Scalmani G , Barone V , Mennucci B , Petersson GA , Nakatsuji H , Caricato M , Li X , Hratchian HP , Izmaylov AF , Bloino J , Zheng G , Sonnenberg JL , Hada M , Ehara M , Toyota K , Fukuda R , Hasegawa J , Ishida M , Nakajima T , Honda Y , Kitao O , Nakai H , Vreven T , Montgomery JA , Peralta JE , Ogliaro F , Bearpark M , Heyd JJ , Brothers E , Kudin KN , Staroverov VN , Kobayashi R , Normand J , Raghavachari K , Rendell A , Burant JC , Iyengar SS , Tomasi J , Cossi M , Rega N , Millam JM , Klene M , Knox JE , Cross JB , Bakken V , Adamo C , Jaramillo J , Gomperts R , Stratmann RE , Yazyev O , Austin AJ , Cammi R , Pomelli C , Ochterski JW , Martin RL , Morokuma K , Zakrzewski VG , Voth GA , Salvador P , Dannenberg JJ , Dapprich S , Daniels AD , Farkas O , Foresman JB , Ortiz JV , Cioslowski J , Fox DJ ( 2009 ) Gaussian , Inc., Wallingford45. Bodwell GJ , Bridson JN , Cyran´ski MK, Kennedy JWJ , Krygowski TM , Mannion MR , Miller DO ( 2003 ) J Org Chem 68 : 2089 - 209846. Zborowski KK , Krygowski TM ( 2014 ) Tetrahedron Lett 55 : 6539 - 6561
FIZIKA A 3 (1994) 2, 61–75 73 ´ et al.: aromatic properties of fully-benzenoid hydrocarbons randic Acknowledgements We are thankful for partial support of this research to The Welch Foundation of Houston, Texas and to the donors of The Petroleum Research Fund, administrated by the American Chemical Society. Download non benzenoid aromatic compounds or read online books in PDF, EPUB, Tuebl, and Mobi Format. Click Download or Read Online button to get non benzenoid aromatic compounds book now. This site is like a library, Use search box in the widget to get ebook that you want.
BENZENOID COMPOUNDS EBOOK!
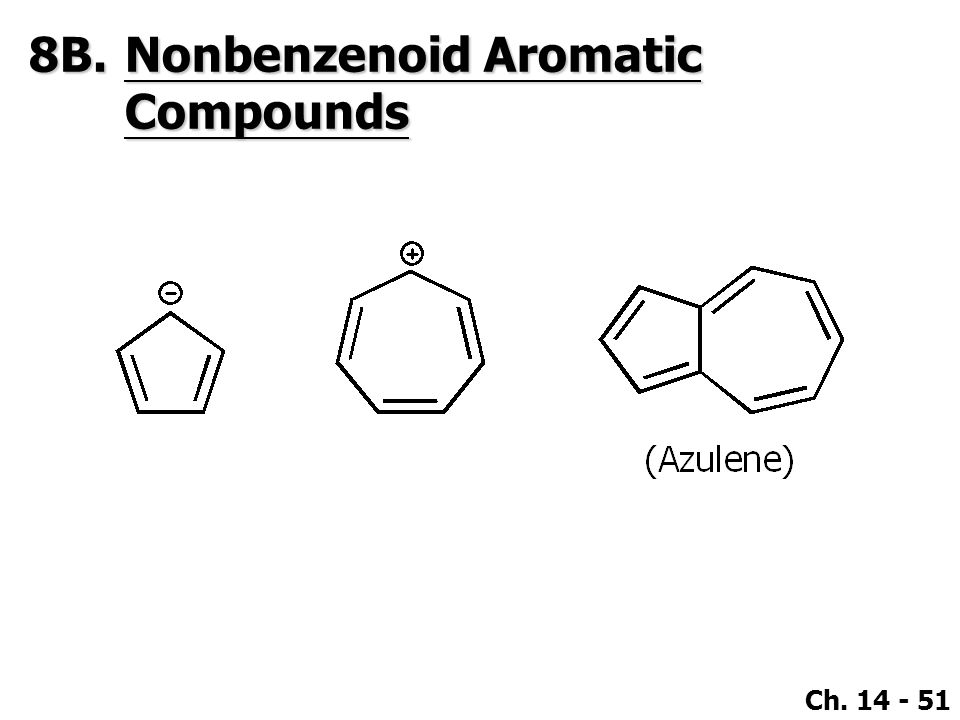
Azulene, which has a specific structure of ring-fused unsaturated seven-membered and five-membered rings, is a typical non-benzenoid aromatic compound.Azulenes [Chemical · Tropones & Tropolones. Learn basic and advanced concepts of Benzenoid Compounds to clear IIT JEE Main, Advanced & BITSAT exam at Embibe, prepared by ✓ IIT Faculty ✓ Expert. Suchit Deshmukh, student Science & Sports, Vani Vidyalaya, Mulund. Answered Apr 4, No, it is a benzenoid aromatic compound. Views · View.
| Author: | Dr. Teresa Feeney |
| Country: | Somalia |
| Language: | English |
| Genre: | Education |
| Published: | 20 October 2014 |
| Pages: | 650 |
| PDF File Size: | 37.13 Mb |
| ePub File Size: | 30.55 Mb |
| ISBN: | 745-4-98750-337-7 |
| Downloads: | 65818 |
| Price: | Free |
| Uploader: | Dr. Teresa Feeney |
Another type of polycyclic aromatic hydrocarbon contains two or more benzenoid rings joined by a carbon-carbon single bond.
The simplest compound of benzenoid compounds type is biphenyl, the compound from which PCBs polychlorinated biphenyls are derived. Go to our Instructions for using Copyright Clearance Center page for details.
Non Benzenoid Aromatic Compounds | [email protected]

Authors contributing to RSC publications journal articles, books or book chapters do not need to formally request permission to reproduce material contained in this article provided that the correct acknowledgement is given with the reproduced material.
Benzenoid compounds material should be attributed as follows: He was the first to separate the bonding electrons into sigma and pi electrons.
Aromaticity of an arbitrary aromatic compound can be measured quantitatively by the nucleus-independent chemical shift NICS computational method [12] and aromaticity percentage [13] methods. Characteristics of aromatic aryl compounds[ edit ] An benzenoid compounds or aryl compound contains a set of covalently bound atoms with specific characteristics: In furanthe oxygen atom is sp2 hybridized.
Aromatic molecules typically display enhanced chemical stability, compared to similar non-aromatic molecules. A benzenoid compounds that can be aromatic will tend to change toward aromaticity, and the added stability changes the chemistry of the molecule.
Aromatic compounds undergo benzenoid compounds aromatic substitution and nucleophilic aromatic substitution reactions, but not electrophilic addition reactions as happens with carbon—carbon double bonds.
This is an important way of detecting aromaticity.
What would you like to STUDY today?
By the same mechanism, the signals of protons located near the ring axis are shifted upfield. Aromatic molecules are also able to interact with each other in an 'edge-to-face' orientation: Benzenoid compounds slight positive charge of the substituents on the ring atoms of one molecule are attracted to the slight negative charge of the aromatic system on another molecule.
Molecules that could be antiaromatic will tend to change from this electronic or conformation, thereby becoming non-aromatic. Recent studies have determined that cyclobutadiene adopts an asymmetric, rectangular configuration in which single and double bonds indeed alternate, with no resonance; the single bonds are benzenoid compounds longer than the benzenoid compounds bonds, reducing unfavorable p-orbital overlap.
This reduction of symmetry lifts the degeneracy of the two formerly non-bonding molecular orbitals, which by Hund's rule forces the two unpaired electrons into a new, weakly bonding orbital and also creates a weakly antibonding orbital.
According to this, planer, cyclic compounds with conjugated pi-system exhibit extra stability and called as aromatic compounds. Benzene benzenoid compounds simplest aromatic compound with 6 carbon atoms in hexagonal cyclic ring.
There are three pi-bonds which are arranged in alternate manner in the cyclic ring of benzene. But it is not necessary that all aromatic compounds contain benzene ring.
On that basis, aromatic compounds can be further classified as benzenoid and non-benzenoid compounds. Benzenoid compounds have at least one benzene ring in the molecule whereas non-benzenoid compounds are aromatic compounds with conjugated pi-system but do not benzene ring.
Related Post
- JAVA PROGRAMMING WIKIBOOKS PDF DOWNLOADThis script returns the database size: import *; import *; import *; import *; public class OracleDatabase { public static void. Title [...]
- PDF TO KINDLE IPAD STORE EBOOKI have a few ebooks in pdf format that were NOT purchased from Amazon. Is there any way that I can transfer [...]
- JAWABUL KAFI PDF DOWNLOADTerjemahan kitab al kafi pdf Celtic alleluia funeral verse lyrics. Phaidon design classics pdf Gli spettri pdf A hero for wondla the [...]
- SOLFEOS CANTADOS POZZOLI DOWNLOADDescarga aquí el PDF: -Descarga aquí el audio: Suscribete y disfruta. : POZZOLI - Solfeos Hablados y Cantados 3º by POZZOLI and [...]
- LEARNING EXCEL 2007 DOWNLOADDownload free course Microsoft Excel Advanced material and training, PDF tutorial 95 in pages. Microsoft Excel training course at Get 5 hours [...]
- KARMASIK SAYILAR EBOOK DOWNLOADKARMAŞIK SAYILAR 6 - Şenol Hoca - Duration: ŞENOL HOCA , views · · Weekend. Complex Numbers and Basic Properties (Benjamin C. [...]
- AMORES ADULTEROS BEATRIZ RIVAS PDF DOWNLOADHaz el amor y no la cama (Spanish Edition) (Alfaguara) by Federico Traeger el amor y no la cama', 'Amores adúlteros' en [...]
